
Continuous Direct Compression Symposium event
Wed, 24 Apr 2024
Event Details
Join us for the 'Continuous Direct Compression' Symposium hosted by CPI’s Medicines Manufacturing Innovation Centre as we delve into the benefits of CDC technology and its future applications.
Oral solid dose medication accounts for 90% of the global pharmaceutical formulations for human use, and demand is increasing due to a naturally aging population. Increased longevity leads to more people living with chronic conditions, and this increases the demand for daily medication.
Continuous direct compression (CDC) of oral solid dosage medicine is a novel manufacturing method that aims to eliminate some of the cost and complexity of old traditional batch processes.
Combining CDC with digital, data-led capabilities can make it even more efficient, adaptable and more sustainable.
Although CDC is a promising technology, it has yet to fulfil its potential in terms of market penetration. With the first product commercially approved in 2015 and only a handful of others following suit, the pharmaceutical industry needs to understand the full operational gains from converting a traditional batch manufacture to fully embrace the benefits of one that is continuous.
In terms of regulatory engagement, the early signs are positive. Regulators are engaged in this topic, but there are issues with a standardised approach. Although the publication of the ICH Q13 guidelines for continuous manufacturing signals a coordinated approach, physical and digital capability are not streamlined across the sector, creating a gap in regulatory understanding which is currently addressed on a case-by-case basis.
Come and engage with us, as we bring key stakeholders together from across the sector to discuss;
- Can we, as a pharmaceutical industry, truly claim to understand the benefits of CDC technology?
- How do we drive forward the successful uptake and deconstruct preconceived barriers to CDC adoption?
- What are the wider industrial experiences that we can share to increase valuable knowledge?
Featuring speakers from Hovione, AstraZeneca, Applied Materials and Gericke, with a variety of pertinent backgrounds, including Commercial Manufacturing, Research and Development, Software providers and PAT specialists…..it will certainly be an eventful discussion!
Speaker overview
Find out more about our speakers for this event
- Filipe Gaspar – Hovione – VP Technology Intensification
- Sean Clifford – AstraZeneca – Senior Process Engineer
- Richard Elkes – GSK – Drug Product Team Leader
- Ewan Norton – MHRA – Lead Senior GMDP inspector (panel member)
- Jason Fung – Applied Materials – Engineering Consultant
- James Kimber – Pfizer – Principal Scientist
- Bernhard Meir – Gericke — Head of Continuous Manufacturing
To register for this event, simply fill out the form below.
If you have any questions about this event, please feel free to contact our marketing team at events@uk-cpi.com
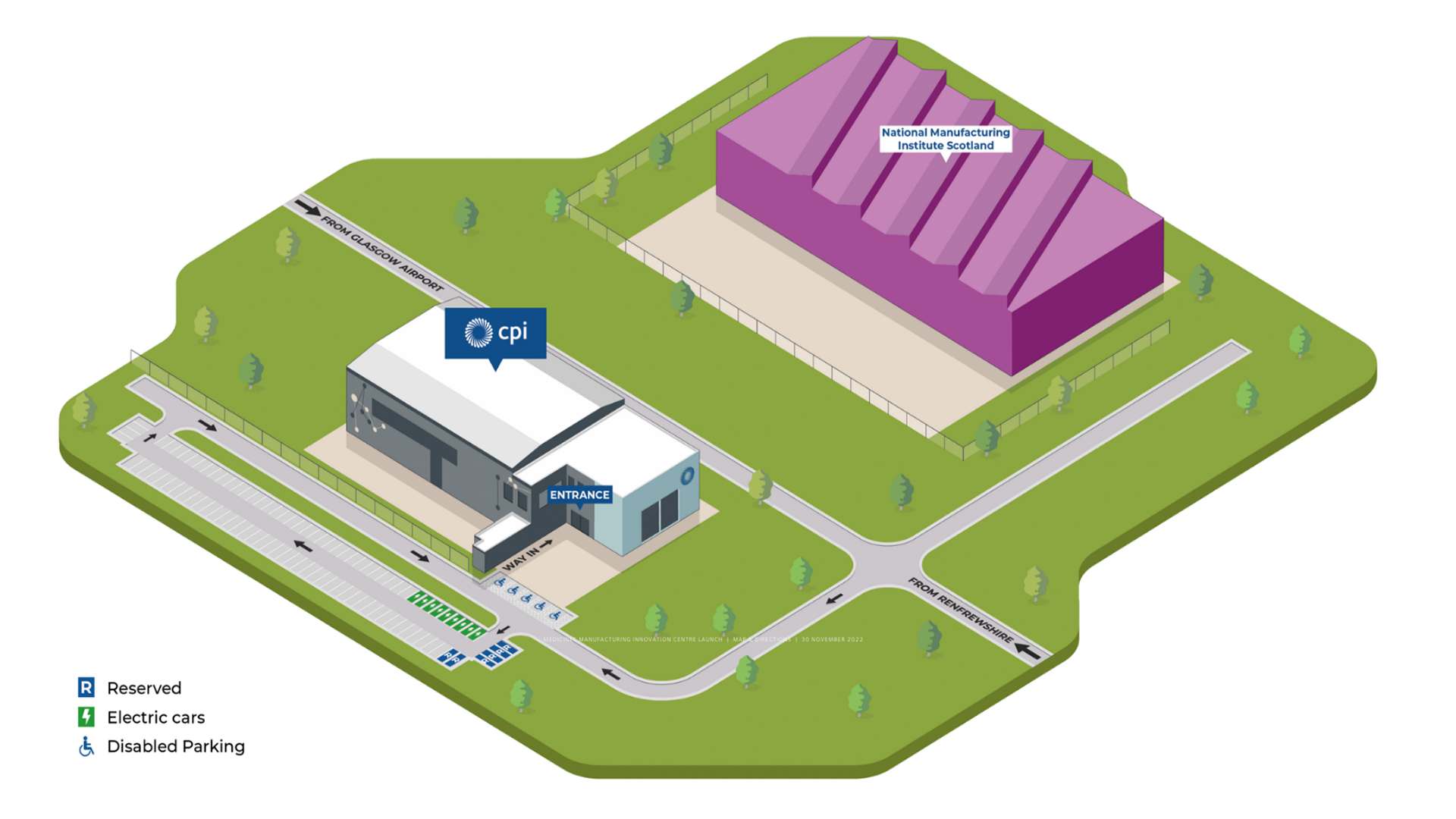
Directions to the Medicines Manufacturing Innovation Centre
CPI’s Medicines Manufacturing Innovation Centre is located in the Advanced Manufacturing Innovation District Scotland (AMIDS), adjacent to Glasgow Airport, at 1 Netherton Square, Paisley, Glasgow, PA3 2EF.
Travelling by car
The postcode is PA3 2EF, and there is free onsite car parking.
Click here to view directions to the Medicines Manufacturing Innovation Centre.
Directions: from M8 motorway both west and eastbound, exit at junction 28 for Glasgow Airport, this will take you onto Sanderling Road. At the roundabout take the second exit.
You will approach a second roundabout, take the first exit onto Abbotsinch Road. Then at the first roundabout, take the second exit, continue straight and you will approach a T junction, turn right, and the Medicines Manufacturing Innovation Centre will be on your right hand side.
Public transport
There are regular trains from Glasgow Central Station to Paisley Gilmour Street Station. From here take a taxi of approx. 5 – 10 minutes to the Medicines Manufacturing Innovation Centre site.
Hotels
We would recommend the Maldon Hotel in Glasgow city centre and the Courtyard by Marriott close to our facility.